An Intuitive Platform for Optimized Clinical Protocol Design
Clinical trial leaders make critical decisions based on spreadsheets, instincts, outdated tools, and dozens of meetings and conversations.
Faro’s technology streamlines this process so that teams can make faster, better-informed decisions about protocol design.
Protocol Design
Optimize Study Design with Data-Driven Insights
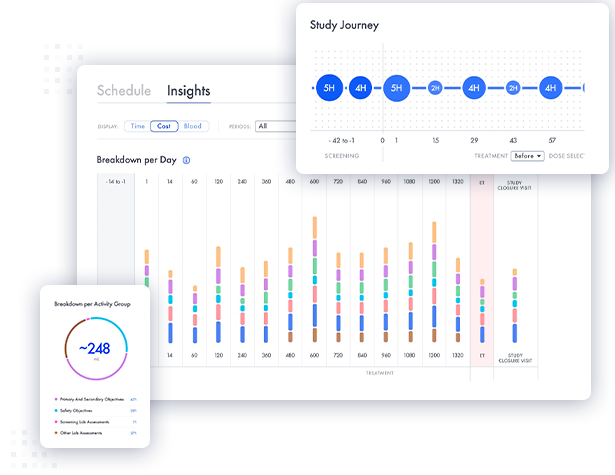
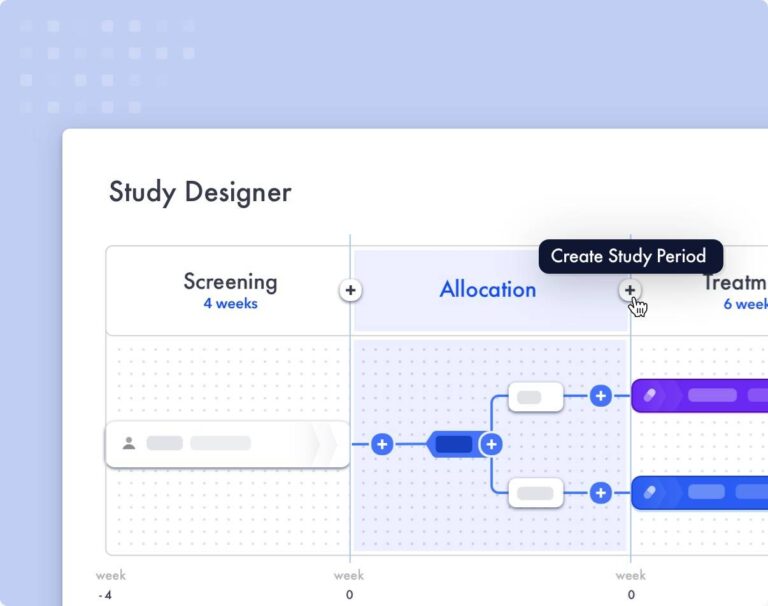
Faro’s Study Designer enables clinicians and scientists to optimize clinical study designs, significantly reducing cost, patient burden and other key trial performance measures. With minimal training, teams can create and configure study-defined procedures, and access real-world metrics that allow them to see the impact of design choices. This enables clinicians and scientists to dynamically modify the Schedule of Activities based on real-time feedback to optimize study requirements. Additional features like procedure grouping, patient burden assessments, and within-visit schedules enable in-depth protocol fine tuning to ensure a trial design that maximizes feasibility and patient-friendliness while reducing costs and site burden.
Collaborate in Real Time
The Faro Study Designer empowers your team to seamlessly collaborate on clinical trial designs with confidence, guided in real time by data-driven insights. Users can explore various iterations of trial designs and assess their performance metrics, gaining quantitative insights into the impact of their design changes. By enabling synchronous teamwork, your team can adeptly address challenges, rapidly iterate on design changes, and streamline processes to minimize both time and costs.
Navigate Complexity with Ease
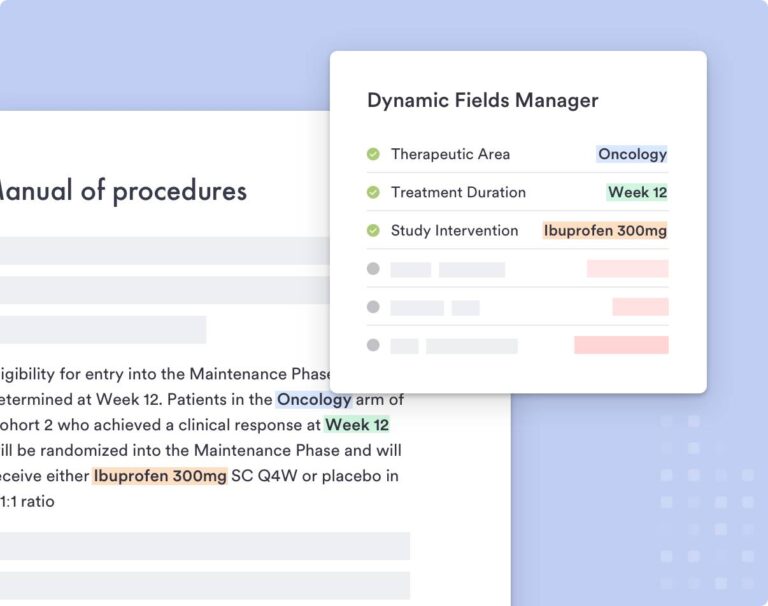
The Faro Study Designer addresses the requirements of modern complex trial design elements by converting them into easily comprehensible standardized content. As narrative components of the protocol are generated in the Study Designer, you can utilize crucial elements as reusable content to promote standardization and efficiency within or across clinical programs.
AI Copilot
Clinical Trial Protocol Generation
Clinical trial protocol documents are highly complex and AI models must be carefully tuned and prompted in order to produce high-quality, complete supportive output. Faro combines the power of the detailed, structured clinical trial model that underlies its Study Designer with the generative capabilities of large language models (LLMs) to produce clinical trial protocol documentation in a fraction of the time and cost of authoring them manually. These documents can be quickly regenerated as the trial design changes, bringing further time and cost savings.
Additional Downstream Document Generation
Faro is constantly adding new clinical documentation to the roster of generated content it supports. Later this year, the AI Copilot will automatically assemble informed consent forms that factor in details from the clinical trial such as footnotes, assessments, and other data that help ensure the form is accurate and complete.
Clinical Outcome Prediction
Faro is applying a range of machine learning models to the problem of clinical trial outcome prediction. The company plans later this year to expand the insights it currently provides on trial performance and cost to include the likelihood of key future trial outcomes. This includes factors such as cycle times or how many amendments the trial design is likely to require. Applying AI to trial insights will further compound the cost and time savings that Faro can bring to clinical trial development across the lifetime of the trial.
Downstream System
Integration & Digital Data Flow
Unlock Study Information Potential
Faro Health helps you optimize your protocol design to fit the complexities of the entire clinical development workflow. A digital-first protocol built with the Faro Health platform allows you to easily utilize all study design information across the clinical continuum.
Enable Downstream Automation with Versatile Content Elements
Faro supports the output of structured, versatile protocol design elements in document format by seamlessly populating common clinical templates that enable downstream automation, including TransCelerate’s Common Protocol Template (CPT) or ICH M11: Clinical electronic Structured Harmonised Protocol (CeSHarP) formats. Users can create and curate reusable content elements during protocol design that easily adapt to other key documents, from the Statistical Analysis Plan (SAP) to the Clinical Study Report (CSR) and beyond.
Align with Industry Data Standards
Faro’s information model also connects structured protocol elements to industry data standards. Study design components, including the Schedule of Activities, can be easily configured for use in downstream data collection systems.
Ready to see Faro in action?
Set up a call to learn more about transforming your clinical trial protocol design.